
Veterinary PRP Preparation System
Platelet-Rich Plasma Preparation System for Veterinary Practices
Designed for Veterinary PRP Preparation
Exclusively from Transcend Biologics
TropoVet® PRP preparation systems include components engineered to support the separation and concentration of platelets from whole blood.
Key system features include:
• Sterile single-use PRP preparation kits
• Gel separator technology for platelet concentration
• Proprietary anti-coagulant for physiologic pH
• Internally coated glass to optimize platelet capture
• Designed for in-clinic point-of-care preparation
These features support a reliable PRP preparation process for veterinary professionals.

PRP Preparation Technology for Equine Practices
Equine veterinarians working with performance horses, rehabilitation programs, and specialty veterinary practices often evaluate PRP preparation systems designed for larger blood volumes and efficient platelet concentration.
The new TropoVet PRP 40 mL system is designed to support these needs by providing a preparation workflow suitable for equine veterinary applications.
Veterinary professionals using PRP preparation systems may include:
• equine veterinarians
• veterinary sports medicine specialists
• equine rehabilitation centers
• veterinary orthopedic practices
• equine performance clinics

TropoVet® PRP System Highlights
The TropoVet® system is designed to support consistent platelet concentration and reproducible preparation workflows.
Key characteristics include:
- nearly 0% red blood cells
- leukocyte-poor PRP profile
- physiologic pH
- simple vacuum blood draw
- reproducible preparation workflow
- consistent performance across procedures
- efficient platelet concentration workflow
- up to 4.7× platelet concentration in a single spin
Simple PRP Preparation Workflow
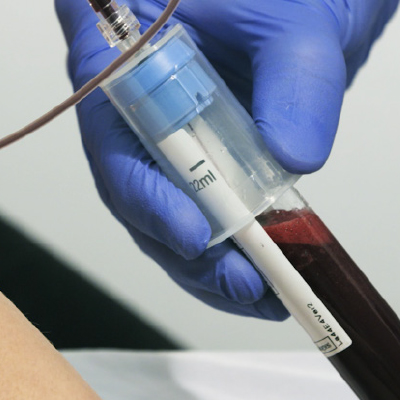
Collect Patients Blood
11, 22, and 40 mL Tube Sizes

Centrifuge (10 min.)

Extract Platelet-Poor Plasma (PPP)
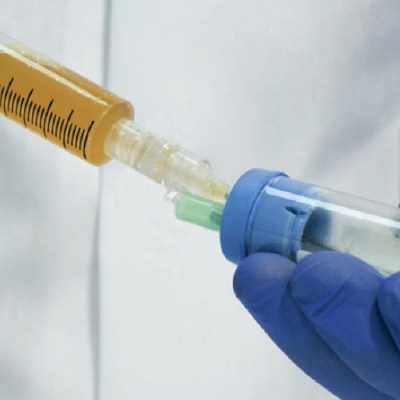
Collect Platelet-Rich Plasma (PRP) and apply
TropoVet® PRP System Specifications
TropoVet® PRP preparation systems are designed for veterinary professionals seeking flexible platelet-rich plasma preparation options across a range of blood volumes. Available in 11 mL, 22 mL, and 40 mL kit configurations, the system supports efficient platelet concentration using a defined preparation workflow.
These kit sizes allow veterinary practices to select a preparation format aligned with patient size, workflow preferences, and target PRP output.
Why TropoVet® PRP
TropoVet® PRP makes high concentration Platelet-Rich Plasma (PRP) preparation simple and reproducible for every medical practice. In a single spin, the system can isolate 80% (+/- 10%) of the platelets while removing the majority of red blood cells and neutrophils.
High Concentration
80% (+/- 10%) Platelet Yield + customizable concentrations up to 4.5x
Safe & Autologous
FDA Cleared 510(k) Class II Medical Device (BK110035) Non-Pyrogenic – Sterile – Closed System
Monocyte Solution
System produces PRP in which monocytes outnumber neutrophils
Simple, Quick, & Predictable
Small blood draw, easy to use, delegatable, reproducible collection process
Low Inflammation
Eliminates nearly 100% of red blood cells and 95% of white blood cells
Comfortable
Proprietary MNC7 anti-coagulant produces physiologic pH, which reduces irritation

Safety and Manufacturing Standards
TropoVet® PRP preparation systems are manufactured under controlled quality standards.
Production environments include:
- ISO 7 cleanroom manufacturing
- critical processes performed in ISO 5 environments
- manufacturing facilities that follow ISO 13485 certification standards
- FDA-registered production facilities
Independent third-party testing includes:
- delayed hypersensitivity testing (GMPT)
- cytotoxicity testing by elution
- acute systemic toxicity testing
More than 850,000 clinical uses worldwide have been reported since 2013.
Clinicians may request testing documentation through their Transcend Biologics representative.

Precautions
Clinicians should observe the following precautions:
- Apply PRP only to the patient from whom the blood was drawn.
- Do not store tubes below 0°C, as breakage may occur.
- Do not store tubes above 40°C, as gel stability may be affected.
- Do not use tubes if foreign matter is present.
- Do not use tubes past their expiration date.
- Do not re-sterilize or attempt to clean the product.
- Do not use products if the Tyvek® package is opened or damaged.
Potential Risks
As with any clinical procedure, potential risks may include:
- infection
- bleeding
- nerve damage
- pain
- lack of clinical response
Clinicians should follow appropriate medical judgment and product instructions.
.
Regulatory Status
PRP preparation systems are classified as Class II medical devices by the U.S. Food and Drug Administration (FDA).
Products sold in the United States must obtain FDA 510(k) clearance before being marketed for PRP preparation.
TropoVet® PRP is cleared under 510(k) number BK110035.
The system is intended for the preparation of autologous platelet-rich plasma from a small blood sample at the patient point of care.
Clinicians may mix the prepared PRP with autograft or allograft bone prior to application to a bony defect to improve handling characteristics.
Reference:
Beitzel K, Allen D, Apostolakos J, et al.
US definitions, current use, and FDA stance on platelet-rich plasma in sports medicine.
Journal of Knee Surgery. 2015.
Request Information About TropoVet® PRP
Healthcare professionals who want to learn more about the TropoVet® PRP preparation system can contact Transcend Biologics for product information, technical specifications, and availability.
Our team can also discuss how PRP preparation technology may integrate into medical practice workflows.