Tropocells® PRP System
The Tropocells PRP system helps physicians prepare platelet-rich plasma (PRP) with a simple and consistent process.
First, clinicians collect a patient’s blood. Next, they spin the sample in a centrifuge. Then, the system separates the blood and concentrates platelets in the plasma.
Because PRP comes from the patient’s own blood, it is an autologous biologic. In addition, the system uses sterile, single-use parts and clear spin settings. As a result, clinicians can prepare PRP in their office with a steady and repeatable method.

Designed for Consistent Results
The system uses engineered components to support reliable platelet separation and consistent preparation output for clinicians preparing PRP at the point of care.
In addition, the system’s design helps reduce variation between procedures. As a result, teams can follow a more predictable process.
Key features include:
- sterile, single-use kits
- gel separator for platelet concentration
- proprietary anticoagulant for patient comfort
- anti-stick coating to improve platelet capture
- point-of-care workflow
Together, these features support a simple and efficient process.
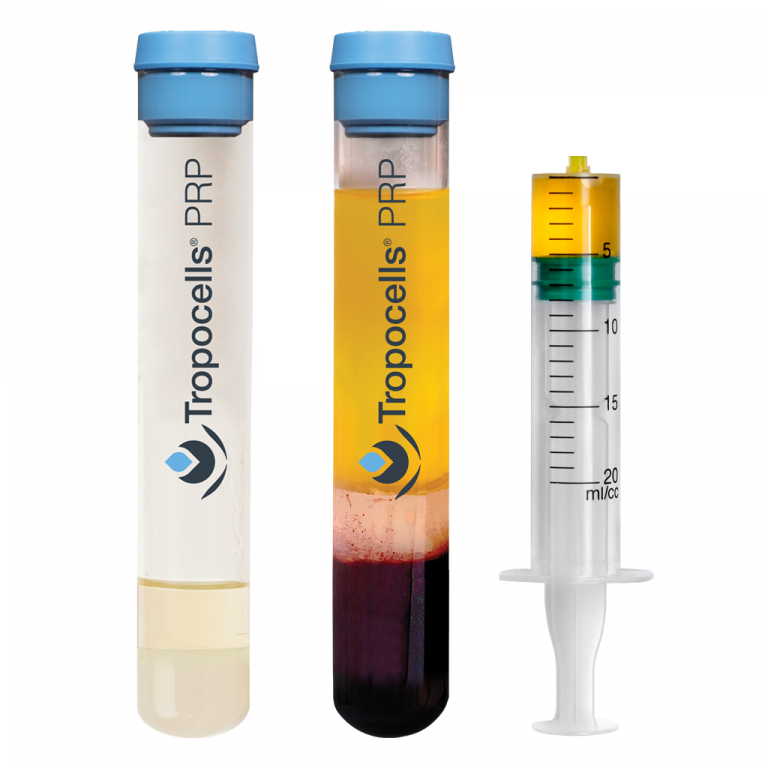
Tropocells PRP System Highlights
The system supports steady platelet concentration and repeatable workflows. In addition, it helps simplify PRP preparation in clinical settings.
Because of this, clinicians can focus more on patient care and less on process variation.
Key characteristics include:
- nearly 0% red blood cells
- leukocyte-poor PRP profile
- physiologic pH
- simple vacuum blood draw
- consistent performance
- efficient platelet concentration
- up to 4.7× concentration in one spin
Designed for Clinical Use
Clinicians across many specialties use PRP systems. For example, the Tropocells system fits into:
- orthopedic practices
- sports medicine clinics
- wound care clinics
- outpatient surgical practices
- veterinary medicine
Because these teams already perform injections and blood draws, they can add PRP without major workflow changes. As a result, adoption is straightforward.

Simple PRP Preparation Workflow
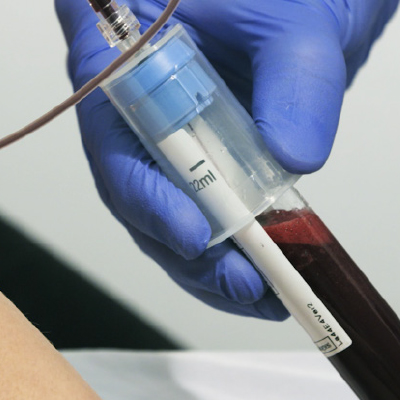
Collect Patients Blood
(11 mL, 22 mL, or 40 mL)

Centrifuge (10 min.)

Extract Platelet-Poor Plasma (PPP)
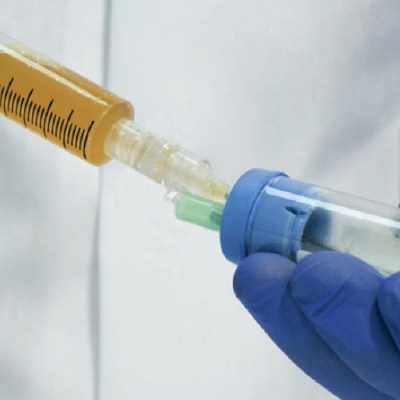
Collect Platelet-Rich Plasma (PRP) and apply
Why Clinicians Choose Tropocells® PRP
The Tropocells PRP system supports a simple and predictable process. In addition, it helps improve efficiency in clinical settings.
Because of this, many clinicians choose it for routine PRP preparation.
High Platelet Concentration
80% (+/- 10%) Platelet Yield + customizable concentrations up to 4.5x
Safe & Autologous
FDA Cleared 510(k) Class II Medical Device (BK110035) Non-Pyrogenic – Sterile – Closed System
Monocyte-Dominant profile
System produces PRP in which monocytes outnumber neutrophils
Simple and Predictable Workflow
Small blood draw, single centrifuge spin, reproducible preparation method
Reduced Red Blood Cell Content
Eliminates nearly 100% of red blood cells and 95% of white blood cells
MNC7 Anticoagulant
The proprietary MNC7 anticoagulant is formulated to maintain pH within a defined range during the centrifugation process, supporting consistent platelet separation in the prepared sample.
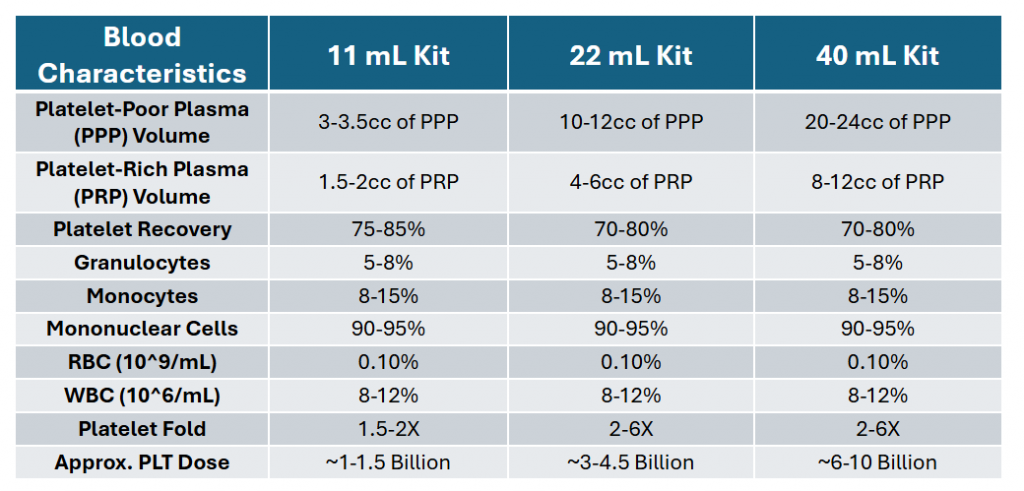
System Specifications
The system offers multiple kit sizes. Therefore, clinicians can choose what fits their needs.
- 11 mL – low volume
- 22 mL – mid range
- 40 mL – higher volume
Each option supports consistent separation using the same process. As a result, workflow stays consistent across cases.


Safety and Manufacturing Standards
Tropocells® PRP systems follow strict quality standards. In addition, manufacturing occurs in controlled environments.
Production includes:
- ISO 7 cleanroom environments
- ISO 5 critical processing areas
- ISO 13485-certified facilities
- FDA-registered manufacturing
Independent testing includes:
- hypersensitivity testing
- cytotoxicity testing
- systemic toxicity testing
Tropocells® PRP systems have been distributed to medical and veterinary practices across the United States since 2013.
Regulatory Status
Tropocells® PRP preparation systems are Class II medical devices regulated by the FDA under 21 CFR Part 864. These systems are intended for processing autologous blood at the point of care. Clinical applications and outcomes are determined solely by the treating healthcare provider. Tropocells® PRP systems are not intended to diagnose, treat, cure, or prevent any disease or medical condition.”
Tropocells® PRP is cleared under BK110035. In addition, the system prepares PRP at the point of care using a small blood sample.
Request Information
Healthcare professionals can contact Transcend Biologics for:
- product information
- technical specifications
- availability
In addition, our team can explain how PRP fits into your workflow. Therefore, you can evaluate whether it meets your practice needs.